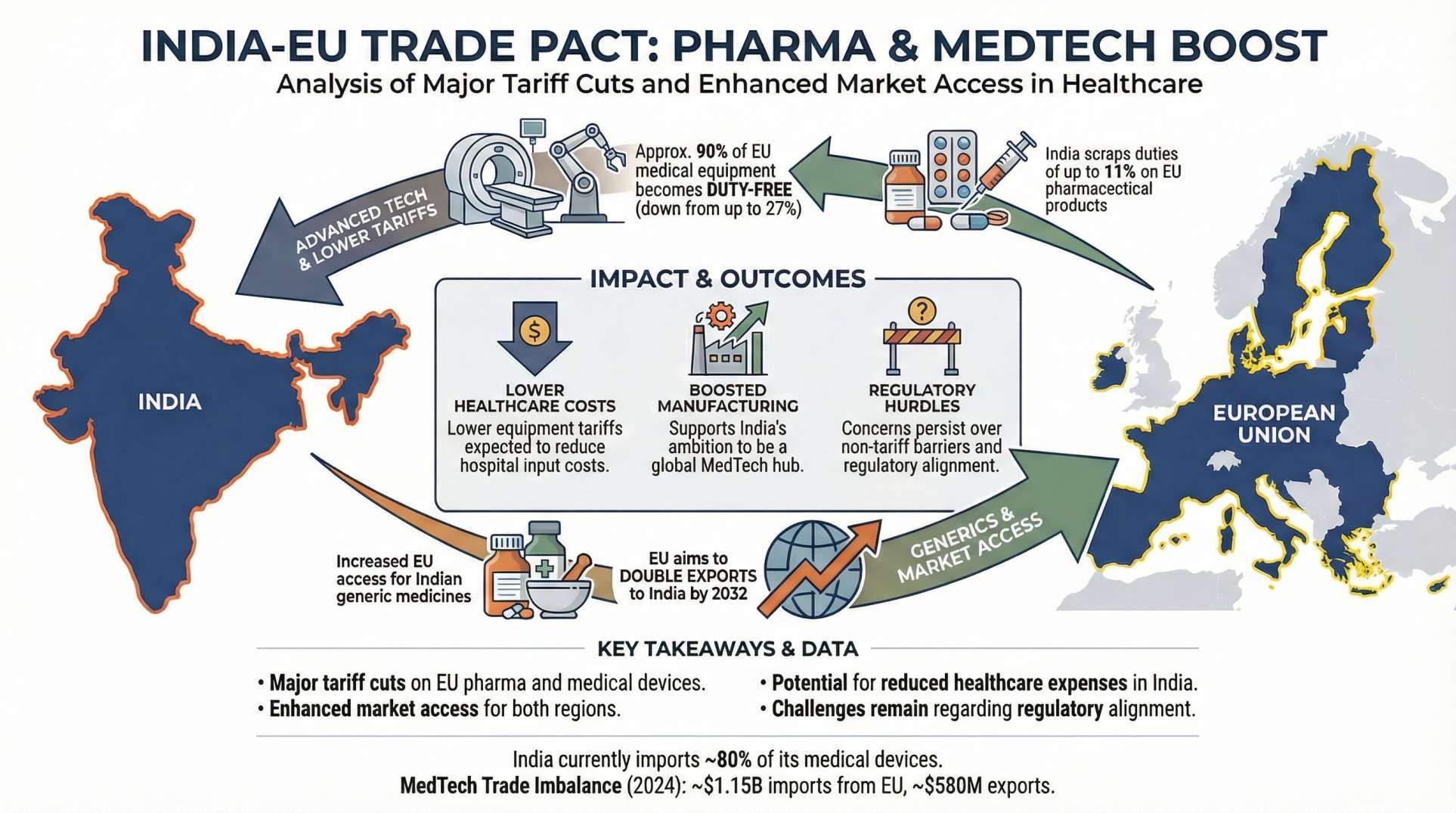
The newly finalised Free Trade Agreement (FTA) between India and the European Union is poised to give a major boost to India’s pharmaceutical and medical devices industries by opening access to the EU’s $572.3 billion healthcare market, government officials say.
📈 Opportunity for Growth
Under the agreement, Indian drug makers and medical technology companies will gain preferential access to one of the largest and most regulated healthcare markets in the world — potentially accelerating export growth and cementing India’s role as a global manufacturing hub.
According to India’s Ministry of Chemicals and Fertilizers, the deal is expected to:
- Boost exports of pharmaceuticals and medical devices to the EU.
- Expand employment opportunities and strengthen participation by MSMEs (micro, small, and medium enterprises).
- Deepen integration into global supply chains, reinforcing India’s image as a reliable healthcare manufacturing partner.
The pact aligns with India’s ongoing ambition to grow its healthcare manufacturing ecosystem and broaden international market reach.
🏭 Beyond Export Access
While the FTA removes or reduces tariffs and simplifies market entry, industry experts caution that success will depend on India’s ability to meet Europe’s stringent quality and regulatory requirements. The EU’s market places high emphasis on product consistency, documentation, and compliance across the lifecycle of pharmaceuticals and medtech products.
Officials say capacity expansion, better quality infrastructure, and skilled manpower will be key for Indian firms to convert market access into sustained export growth.